Global Intravenous Immunoglobulin Market
Global Intravenous Immunoglobulin Market Size, Share, By Product Type (IgG, IgA, IgM, IgD, IgE), By Application (Immunodeficiency diseases, Chronic Inflammatory Demyelinating Polyneuropathy (CIDP), Myasthenia Gravis, Multifocal Motor Neuropathy, Idiopathic Thrombocytopenic Purpura (ITP), Inflammatory Myopathies, Guillain- Barr� Syndrome), By End Use (Hospitals and clinics, Ambulatory surgical centers, Homecare settings), and By Region (North America, Europe, Asia-Pacific, Latin America, Middle East & Africa), Analysis and Forecast 2026-2035
Report Overview
Table of Contents
Market Snapshot
- Global Intravenous Immunoglobulin Market Size (2025): USD 16.5Billion
- Projected Global Intravenous Immunoglobulin Market Size (2035): USD 36.1Billion
- Global Intravenous Immunoglobulin Market Compound Annual Growth Rate (CAGR): 8.14%
- Largest Regional Market: North America
- Fastest Growing Region: Asia-Pacific
- Market 3rd Largest Region: Europe
- Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
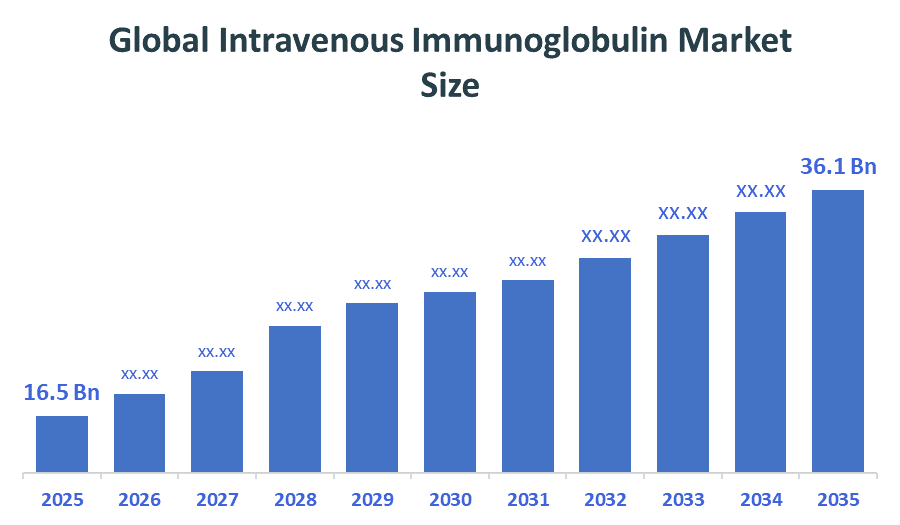
According to Decision Advisors, the Global Intravenous Immunoglobulin Market Size is expected to grow from USD 16.5billion in 2025 to USD 36.1billion by 2035, at a CAGR of 8.14% during the forecast period 2026-2035. The global Intravenous Immunoglobulin Market is projected to grow significantly over the next decade, driven by higher diagnosis rates, new product approvals, a shift toward home-based infusion therapies, rising prevalence of immunodeficiency diseases, expanding therapeutic applications, increased plasma collection, adoption of home infusion therapy, and approval of new products.
Market Overview/ Introduction
The Global Intravenous Immunoglobulin (IVIG) Market refers to a business sector that focuses on producing, selling, and using intravenous immunoglobulin for treating immune deficiency diseases, autoimmune disorders, and neurological diseases. The human plasma-derived intravenous immunoglobulin functions as a vital component of the immune system, which helps to enhance and control immune system activities. There is a rapid expansion within the intravenous immunoglobulin market owing to a growing incidence rate of both primary and secondary immune deficiency diseases, along with autoimmune diseases and other chronic illnesses. The healthcare system receives advantages through new plasma collection methods and manufacturing advancements because these developments enable better patient product delivery, which creates ongoing market expansion through reimbursement systems and home care services.
- The Department of Pharmaceuticals invited applications for critical drug intermediates and APIs under its incentive scheme. It provided financial incentives of 5% to 10% on incremental sales, encouraging companies to manufacture biologics, including IVIG, and strengthening domestic pharmaceutical production in India.
- The act proceeded through the legislative phase in 2026 and aimed to streamline regulatory procedures by targeting 180-day drug approvals. It helped Europe enhance competitiveness with the U.S. and Asian markets while improving efficiency, accelerating market access, and supporting pharmaceutical innovation.
Notable Insights: -
- North America is anticipated to hold the largest share of approximately 57.9% in the intravenous immunoglobulin market over the forecast period.
- Asia Pacific is expected to grow at a rapid CAGR of approximately 9.95% in the intravenous immunoglobulin market during the forecast period.
- The IgG segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 7.9% during the forecast period.
- The immunodeficiency diseases segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 8.1% during the forecast period.
- The hospitals and clinics segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 7.8% during the forecast period.
- The compound annual growth rate of the Global Intravenous Immunoglobulin Market is 8.14%.
- The market is likely to achieve a valuation of USD 36.1billion by 2035.
What is role of technology in grooming the market?
Technology plays a vital role in shaping the growth of the global Intravenous Immunoglobulin (IVIG) market by enhancing production efficiency, safety, and treatment outcomes. Advanced plasma fractionation and purification technologies improve yield and ensure high-quality immunoglobulin with minimal contamination risks. Automation and digital monitoring systems streamline manufacturing processes and maintain regulatory compliance. Innovations in cold chain logistics and supply chain management ensure safe storage and global distribution. Additionally, improved infusion devices and the development of subcutaneous immunoglobulin (SCIg) support convenient, home-based care. Data analytics and biotechnology advancements are also enabling better disease diagnosis and personalized treatment approaches, ultimately increasing patient access, optimizing therapy effectiveness, and driving overall market expansion.
Market Drivers
The global Intravenous Immunoglobulin (IVIG) market experiences strong growth because major factors are currently driving the market. The demand for immunoglobulin replacement therapies shows significant growth because more cases of primary and secondary immunodeficiency disorders have been diagnosed. Medical professionals use IVIG as a treatment for both neuromuscular disorders and autoimmune diseases, which include chronic inflammatory demyelinating polyneuropathy and Kawasaki disease. The plasma collection system improvements and the increased rates of donor participation have created better material access, which enables facilities to produce more output. The growing preference for home-based IVIG administration leads to better patient convenience and higher treatment compliance. The leading companies in the market maintain global market growth through their continuous development of manufacturing technologies and their introduction of innovative product formulations.
Restrain
The global Intravenous Immunoglobulin (IVIG) Market faces restraints due to the limited availability of human plasma, the key raw material, which restricts production capacity. High treatment costs and complex manufacturing processes further limit accessibility, especially in developing regions. Additionally, strict regulatory requirements, risk of supply shortages, and potential side effects associated with IVIG therapy hinder overall market growth.
Competitive Analysis:
The report offers the appropriate analysis of the key organizations/companies involved within the global Intravenous Immunoglobulin Market, along with a comparative evaluation primarily based on their product offerings, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Companies in Global Intravenous Immunoglobulin Market
- CSL Behring
- Grifols S.A.
- Takeda Pharmaceutical Company Limited
- Octapharma AG
- Kedrion Biopharma
- Biotest AG
- Baxter International Inc.
- LFB Group
- China Biologic Products Holdings Inc.
- ADMA Biologics Inc.
- Bio Products Laboratory Ltd.
- Shanghai RAAS Blood Products Co., Ltd
Government Initiatives
|
Country |
Key Government Initiatives |
|
Australia |
The National Immunoglobulin Governance Program was updated to improve the management and clinical use of immunoglobulin therapies. It introduced revised access criteria, strengthened supply planning, and enhanced governance frameworks, ensuring appropriate utilization, better patient access, and more efficient distribution of IVIG products. |
|
Europe |
The European Union reached a landmark agreement on the new Pharma Package, reforming pharmaceutical legislation. It aimed to improve drug accessibility, accelerate approvals, strengthen supply chains, and encourage innovation, thereby supporting the development and availability of therapies, including plasma-derived treatments like intravenous immunoglobulin. |
|
Germany |
BloodTrain trained African regulatory representatives at the Paul-Ehrlich-Institut to strengthen oversight of blood and plasma-derived products. The initiative enhanced regulatory capabilities, improved safety and quality standards, and supported better management of blood products, contributing to a more reliable supply chain for therapies like IVIG. |
Study on the Supply, Demand, Distribution, and Market Environment of the Intravenous Immunoglobulin Market
The global Intravenous Immunoglobulin (IVIG) market is characterized by a complex balance of supply, demand, distribution, and market environment. Demand is rapidly increasing due to the rising prevalence of immunodeficiency, autoimmune, and neurological disorders, along with expanding therapeutic applications. On the supply side, IVIG production depends heavily on human plasma, the primary raw material, collected from thousands of donors and processed through advanced fractionation techniques. However, limited plasma availability and lengthy production cycles create supply constraints. Distribution is mainly driven through hospital and specialty pharmacies, ensuring patient access and treatment continuity. The market environment is highly regulated, capital-intensive, and dominated by a few key players, with ongoing technological advancements improving efficiency, safety, and global accessibility.
Price Analysis and Consumer Behaviour Analysis
The Global Intravenous Immunoglobulin (IVIG) Market exhibits a high-cost pricing structure influenced by complex production processes, limited plasma supply, and strong demand dynamics. IVIG therapy remains expensive, with treatment costs ranging from thousands to tens of thousands annually, driven by labor-intensive plasma collection and strict regulatory requirements. Pricing is also affected by reimbursement policies, competition, and patient willingness to pay, leading to regional price variations. From a consumer behavior perspective, demand is primarily driven by the critical need for treating immunodeficiency and autoimmune diseases, making IVIG a necessity-based therapy. However, high costs and limited insurance coverage in developing regions restrict adoption, while in developed markets, strong healthcare support and awareness encourage higher utilization and long-term treatment adherence.
Market Segmentation
The Intravenous Immunoglobulin Market share is classified into product type, application, and end user
- The IgG segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 7.9% during the forecast period.
Based on the product type, the Intravenous Immunoglobulin Market is divided into IgG, IgA, IgM, IgD, and IgE. Among these, the IgG segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 7.9% during the forecast period. The IgG segment dominated due to its high effectiveness in treating immunodeficiency and autoimmune disorders. Its widespread clinical use, strong therapeutic benefits, and high concentration in IVIG products drive demand, supporting consistent growth and increased adoption across global healthcare systems.
- The immunodeficiency diseases dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 8.1% during the forecast period.
Based on the application, the Intravenous Immunoglobulin Market is divided into immunodeficiency diseases, chronic inflammatory demyelinating polyneuropathy (CIDP), myasthenia gravis, multifocal motor neuropathy, idiopathic thrombocytopenic purpura (ITP), inflammatory myopathies, Guillain- Barré syndrome, and other applications. Among these, the immunodeficiency diseases segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 8.1% during the forecast period. The growth of the immunodeficiency diseases segment is growing due to the rising prevalence of primary and secondary immunodeficiency disorders. Increasing diagnosis rates, growing awareness, and the essential need for immunoglobulin replacement therapy are driving demand, supporting strong growth and widespread adoption of IVIG treatments globally.
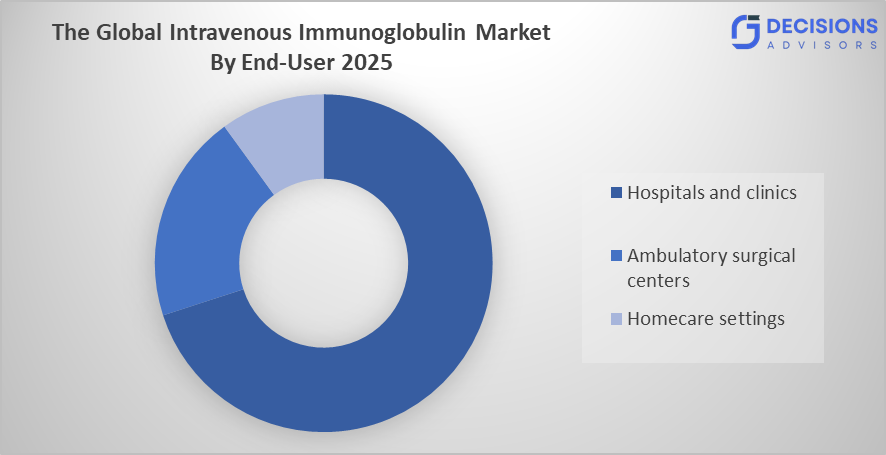
- The hospitals and clinics segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 7.8% during the forecast period.
Based on the end user, the Intravenous Immunoglobulin Market is divided into hospitals and clinics, ambulatory surgical centers, and homecare settings. Among these, the hospitals and clinics segment dominated the market in 2025 and is anticipated to grow at a significant CAGR of approximately 7.8% during the forecast period. The growth of the hospitals and clinics segment is growing due to the availability of advanced healthcare infrastructure, skilled professionals, and proper monitoring facilities for IVIG administration. High patient inflow, better reimbursement support, and preference for clinical supervision during treatment further drive the growth of this segment globally.
Strategies to Implement for Growth of the Market in Non-Leading Regions
To drive growth of the global Intravenous Immunoglobulin (IVIG) market in non-leading regions, companies should focus on expanding plasma collection infrastructure and strengthening local manufacturing capabilities to reduce supply dependence. Strategic partnerships with regional healthcare providers and governments can improve distribution networks and treatment accessibility. Increasing awareness about immunodeficiency and autoimmune disorders through educational campaigns will support early diagnosis and adoption. Additionally, implementing cost-effective pricing strategies and reimbursement support can enhance affordability in emerging economies. Investment in healthcare infrastructure, including infusion centers and trained professionals, is essential. Leveraging alternative delivery methods such as subcutaneous immunoglobulin (SCIg) can further expand access, especially in remote areas, ultimately supporting sustainable market growth.
Regional Segment Analysis of the Intravenous Immunoglobulin Market
- North America (U.S., Canada, Mexico)
- Europe (Germany, France, U.K., Italy, Spain, Rest of Europe)
- Asia-Pacific (China, Japan, India, Rest of APAC)
- South America (Brazil and the Rest of South America)
- The Middle East and Africa (UAE, South Africa, Rest of MEA)
North America is anticipated to hold the largest share of approximately 57.9% the intravenous immunoglobulin market over the forecast period.
North America is anticipated to hold the largest share of approximately 57.9% the Intravenous Immunoglobulin Market over the forecast period. The dominance of this region is primarily driven by its advanced healthcare infrastructure, high awareness, and strong diagnosis rates of immunodeficiency and autoimmune disorders. The region also benefits from well-established plasma collection networks, favourable reimbursement policies, and the presence of leading market players, ensuring high treatment adoption and consistent product availability.
Asia Pacific is expected to grow at a rapid CAGR of approximately 9.95% in the intravenous immunoglobulin market during the forecast period.
Asia Pacific is expected to grow at a rapid CAGR of approximately 9.95% in the Intravenous Immunoglobulin Market during the forecast period. This growth is driven by the improving healthcare infrastructure, rising awareness of immunodeficiency and autoimmune diseases, and increasing diagnosis rates. Growing population, expanding healthcare expenditure, and government initiatives to enhance plasma collection and treatment access are further driving demand, supporting strong market expansion in the region
.
Europe is the 3rd largest region to grow in the intravenous immunoglobulin market during the period.
Europe is the third-largest region in the intravenous immunoglobulin market due to its well-established healthcare systems, increasing prevalence of autoimmune and neurological disorders, and strong regulatory framework. The region also benefits from rising awareness, improving diagnosis rates, and supportive reimbursement policies, which collectively contribute to steady market growth during the forecast period.
Future Market Trends in Global Intravenous Immunoglobulin Market: -
1. Rising Shift Toward Subcutaneous Immunoglobulin (SCIg)
The rising shift toward subcutaneous immunoglobulin (SCIg) is driven by its ability to offer convenient, home-based treatment with fewer hospital visits and reduced healthcare costs. SCIg provides steady immunoglobulin levels, minimizes systemic side effects, and improves patient compliance, especially for chronic conditions. Growing preference for self-administration and advancements in infusion devices are further accelerating its adoption globally.
2. Increasing Demand Driven by Expanding Therapeutic Applications
The increasing demand for intravenous immunoglobulin (IVIG) is driven by its expanding use across a wider range of therapeutic applications, including autoimmune diseases, neurological disorders, and rare immunological conditions. Rising prevalence of chronic illnesses and growing off-label usage are further boosting demand. Continuous clinical research and improved diagnosis are also supporting broader adoption, contributing to sustained market growth in the future.
3. Advancements in Plasma Collection & Biomanufacturing Technologies
Advancements in plasma collection and biomanufacturing technologies are driving future growth in the IVIG market by improving production efficiency and ensuring a stable supply of high-quality immunoglobulins. Enhanced fractionation, purification processes, and automation are increasing yield while maintaining safety standards. Additionally, expanding plasma donation networks and improving storage and logistics systems are helping meet rising global demand effectively.
Recent Development
- In April 2026, GC Biopharma enhanced the safety profile of its intravenous immunoglobulin (IVIG) products by introducing advanced prothrombotic impurity detection technology. The company developed an improved assay to accurately detect Factor XI, reduce thromboembolic risks, and strengthen quality control while aligning with evolving global regulatory standards.
- In March 2025, ADMA Biologics Inc responded to a short-selling report, stating that it had distorted facts and created confusion about the actual efficacy and competitive position of its IVIG product, Asceniv, while reaffirming confidence in the therapy’s performance and market standing.
How is Recent Developments Helping the Market?
Recent developments are significantly strengthening the global intravenous immunoglobulin (IVIG) market by enhancing production efficiency, product quality, and treatment accessibility. Advanced plasma fractionation and purification technologies are improving yield and safety, while high-concentration IVIG formulations are reducing infusion time and increasing patient convenience. Expansion of plasma collection networks is addressing supply shortages and supporting rising demand. Additionally, continuous product innovations, regulatory approvals, and ongoing clinical trials are broadening therapeutic applications, particularly for autoimmune and neurological disorders. The integration of biotechnology and personalized treatment approaches is further improving patient outcomes. Moreover, improved supply chain systems and the emergence of alternative delivery methods like subcutaneous immunoglobulin are increasing accessibility and reducing hospital dependency, collectively driving sustained market growth.
Market Segment
This study forecasts revenue at global, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the Intravenous Immunoglobulin Market based on the below-mentioned segments:
Global Intravenous Immunoglobulin Market, By Product Type
- IgG
- IgA
- IgM
- IgD
- IgE
Global Intravenous Immunoglobulin Market, By Application
- Immunodeficiency diseases
- Chronic Inflammatory Demyelinating Polyneuropathy (CIDP)
- Myasthenia Gravis, Multifocal Motor Neuropathy
- Idiopathic Thrombocytopenic Purpura (ITP)
- Inflammatory Myopathies
- Guillain-Barre Syndrome
Global Intravenous Immunoglobulin Market, By End Use
- Hospitals and clinics
- Ambulatory surgical centers
- Homecare settings
Global Intravenous Immunoglobulin Market, By Regional Analysis
- North America
- US
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- South America
- Brazil
- Argentina
- Rest of South America
- Middle East & Africa
- UAE
- Saudi Arabia
- Qatar
- South Africa
- Rest of the Middle East & Africa
Frequently Asked Questions (FAQ)
Q What are the key challenges in IVIG production?
A. The production of IVIG faces challenges due to its dependence on human plasma, which requires extensive donor participation. The process is time-consuming, highly regulated, and involves complex fractionation and purification steps, making it costly and limiting rapid scalability in response to rising demand.
Q. How does IVIG therapy impact patient quality of life?
A. IVIG therapy significantly improves patient quality of life by strengthening the immune system and reducing the frequency and severity of infections. It also helps manage autoimmune and neurological conditions, enabling patients to lead more stable and productive lives with fewer hospitalizations.
Q. What role do emerging markets play in the IVIG industry?
A. Emerging markets are becoming important growth drivers due to increasing healthcare investments, improving diagnosis rates, and expanding access to advanced therapies. Rising awareness and government support in these regions are creating new opportunities for IVIG adoption and market expansion.
Q. How does regulatory compliance affect the IVIG market?
A. Strict regulatory requirements ensure product safety, quality, and efficacy in the IVIG market. While compliance increases production costs and approval timelines, it builds trust among healthcare providers and patients, ensuring con
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Global |
| Pages | 240 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
