US Amniotic Membrane Market
US Amniotic Membrane Market Size, Share, By Type (Cryopreserved Amniotic Membrane, Dehydrated Amniotic Membrane), By Application (Wound Care, Ophthalmology, Orthopedics, Surgical Applications, Others), By End User (Hospitals, Specialty Clinics, Ambulatory Surgical Centers), Analysis and Forecast 2025?2035
Report Overview
Table of Contents
Market Snapshot
- US Amniotic Membrane Market Size (2025): USD 1.26 Billion
- Projected US Amniotic Membrane Market Size (2035): USD 4.1 Billion
- US Amniotic Membrane Market Compound Annual Growth Rate (CAGR): 12.52%
- Market Base Year: 2025
- Historical Period: 2021–2024
- Forecast Period: 2026–2035
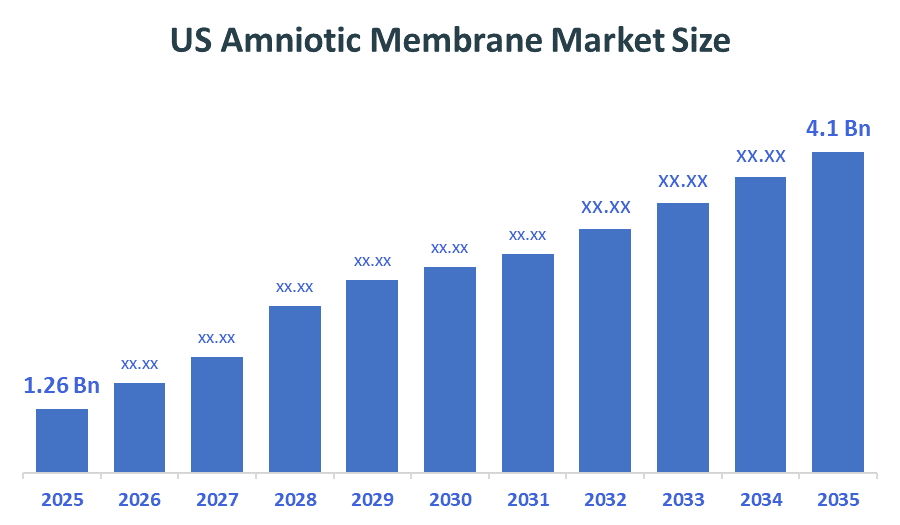
According to Decision Advisors, the US Amniotic Membrane Market Size is expected to grow from USD 1.26 Billion in 2025 to USD 4.1 Billion by 2035, at a CAGR of 12.52% during the forecast period 2025-2035. The US Amniotic Membrane Market is projected to grow significantly over the next decade. The market value is increasing significantly due to rising demand in wound care, ophthalmology, and regenerative medicine applications.
Market Overview/Introduction
The U.S. Amniotic Membrane Market is defined as the use of the innermost placental layer to create biological scaffolds, these membranes are essential for treating everything from chronic diabetic ulcers to complex ocular surface reconstructions. This rapid annual growth is fueled by a perfect storm, an ageing population facing more chronic wounds and a surge in traumatic injuries that require advanced tissue repair. The market’s momentum is anchored by heavy government and corporate investment. Federal support is massive, with the NIH funnelling over 2.5 billion into regenerative research in 2023 alone, while the FDA provides the regulatory clarity needed to bring these tissues safely to clinics. On the private side, industry leaders like MiMedx, Organogenesis, and Integra LifeSciences are locked in a race for innovation. Looking ahead, the future opportunity lies in making these treatments more accessible. They are aggressively pursuing mergers and acquisitions, such as MiMedx’s recent move for Skye Biologics and launching next-gen products like Amnigel Plus to stay ahead of the curve.
- The US reports approximately 1.6 million diabetic foot ulcer cases annually.
Notable Insights: -
- By type, the cryopreserved amniotic membrane segment dominated the market in 2025, accounting for approximately 60% of market share, and is projected to grow at a substantial CAGR during the forecast period.
- By end user, the hospitals segment dominated the market in 2025, accounting for approximately 67% of market share, and is projected to grow at a substantial CAGR during the forecast period.
- The compound annual growth rate of the US Amniotic Membrane Market is 12.52%.
- The market is likely to achieve a valuation of USD 4.1 Billion by 2035.
What is the role of technology in grooming the market?
Technology plays a critical role in enhancing the US amniotic membrane market through innovations in tissue processing, preservation techniques, and product formulations. Advanced cryopreservation and dehydration technologies help maintain biological integrity and extend shelf life. New product formats such as micronized powders and injectable amniotic solutions are improving clinical usability and expanding applications. Additionally, integration with regenerative medicine and stem cell therapies is enhancing treatment outcomes, particularly in chronic wound care and ophthalmology. These advancements have improved healing rates by up to 35% in complex wounds.
Market Drivers
Several factors are fuelling the U.S. market’s growth. At the heart of it is a simple reality, more people are dealing with chronic wounds and undergoing surgery than ever before. With over 17% of Americans now 65 or older, we’re seeing a significant rise in slow-healing injuries and age-related conditions that require specialized care. Doctors are also moving away from basic bandages in favor of biologics like amniotic membranes. On top of that, new FDA pathways and better insurance coverage are making it easier for hospitals and clinics to get these treatments into the hands of the people who need them most. These advanced tissues are a game-changer because they help patients heal faster, lower the risk of infection, and lead to much better long-term results.
Restrain
The U.S. amniotic membrane market faces restraints due to high treatment costs, limited reimbursement coverage, and stringent regulatory requirements. Ethical concerns related to tissue sourcing, variability in product quality, and lack of standardized clinical guidelines further restrict widespread adoption across healthcare providers and limit market growth potential.
Study on the Supply, Demand, Distribution, and Market Environment of the US Amniotic Membrane Market
The US amniotic membrane market demonstrates a well-established ecosystem supported by strong supply chains and regulatory oversight. On the supply side, the market is driven by tissue banks, biotechnology firms, and medical device manufacturers involved in processing and distributing amniotic products. Demand is fueled by increasing cases of diabetic ulcers, burns, surgical wounds, and ocular disorders. Hospitals and outpatient surgical centers are the primary consumers, supported by efficient distribution networks and direct sales channels. The market environment is highly favorable due to advanced healthcare spending, which exceeds USD 4.5 trillion annually in the US, and strong clinical adoption of regenerative therapies. However, regulatory scrutiny and variability in product quality pose moderate challenges.
Price Analysis and Consumer Behaviour Analysis
The pricing of amniotic membrane products varies based on processing type, application, and product form.
- Sheet grafts: USD 500 – USD 3,000 per unit
- Injectable forms: USD 300 – USD 1,500 per treatment
From a consumer behavior perspective, decision-making is largely driven by clinical effectiveness and healing outcomes rather than cost. Physicians prioritize products with proven efficacy, especially for chronic wounds and surgical applications. However, reimbursement coverage plays a crucial role in influencing adoption, particularly in outpatient settings.
Market Segmentation
The US Amniotic Membrane Market share is classified into type, application, and end user.
- The cryopreserved amniotic membrane segment dominated the market in 2025, accounting for approximately 60% of market share, and is projected to grow at a substantial CAGR during the forecast period.
Based on the type, the US amniotic membrane market is divided into cryopreserved amniotic membrane and dehydrated amniotic membrane. Among these, the cryopreserved amniotic membrane segment dominated the market in 2025, accounting for approximately 60% of market share, and is projected to grow at a substantial CAGR during the forecast period. Cryopreserved membranes dominate due to their ability to retain the natural biological properties of the tissue, including growth factors, anti-inflammatory proteins, and structural integrity, making them highly effective in complex wound healing and ophthalmic applications. However, dehydrated amniotic membranes are gaining traction due to their longer shelf life, ease of storage, and convenience in outpatient settings.
- The wound care segment accounted for the largest share in 2025 and is anticipated to grow at a significant CAGR during the forecast period.
Based on the application, the US amniotic membrane market is divided into wound care, ophthalmology, orthopedics, surgical applications, and others. Among these, the wound care segment accounted for the largest share in 2025 and is anticipated to grow at a significant CAGR during the forecast period. Amniotic membranes are extensively used in the treatment of chronic wounds such as diabetic foot ulcers, venous leg ulcers, and pressure ulcers due to their ability to accelerate healing, reduce inflammation, and minimize scar formation. The rising prevalence of diabetes and chronic wounds in the United States is significantly driving this segment. Additionally, the ophthalmology segment is expanding due to increasing use in ocular surface reconstruction and corneal repair procedures.
- The hospitals segment dominated the market in 2025, accounting for approximately 67% of market share, and is projected to grow at a substantial CAGR during the forecast period.
Based on the end user, the US amniotic membrane market is divided into hospitals, specialty clinics, and ambulatory surgical centers. Among these, the hospitals segment dominated the market in 2025, accounting for approximately 67% of market share, and is projected to grow at a substantial CAGR during the forecast period. Hospitals dominate due to the availability of advanced surgical infrastructure, skilled healthcare professionals, and a high volume of patients requiring wound care and surgical procedures. A significant proportion of complex treatments involving amniotic membrane products are performed in hospital settings, reinforcing their leading position. Specialty clinics and ambulatory surgical centers are gradually expanding due to the increasing shift toward outpatient care but remain secondary contributors.
Recent Development
- In March 2026, MiMedx Group, Inc. expanded its product portfolio with the launch of AMNIOFIX thyroid shields, an amniotic membrane-based product designed for endocrine and surgical procedures. This development highlights the growing application of amniotic membrane products beyond traditional wound care into specialized surgical segments.
- In October 2024, Merakris Therapeutics launched Dermacyte AC Matrix, a lyophilized amniotic membrane allograft aimed at advanced wound care and tissue regeneration, expanding treatment options for chronic wounds.
- In June 2024, BioTissue Inc. introduced CAM360 AG, a next-generation cryopreserved amniotic membrane designed for dry eye and ocular surface conditions, offering improved handling and patient comfort.
Competitive Analysis
The report offers the appropriate analysis of the key organisations/companies involved within the US amniotic membrane market, along with a comparative evaluation primarily based on their product offering, business overviews, geographic presence, enterprise strategies, segment market share, and SWOT analysis. The report also provides an elaborative analysis focusing on the current news and developments of the companies, which includes product development, innovations, joint ventures, partnerships, mergers & acquisitions, strategic alliances, and others. This allows for the evaluation of the overall competition within the market.
Top Key Companies in the US Amniotic Membrane Market
- MiMedx Group, Inc.
- Organogenesis Holdings Inc.
- Smith & Nephew plc
- Stryker Corporation
- Integra LifeSciences
Key Target Audience
- Market Players
- Investors
- End-users
- Government Authorities
- Consulting and Research Firm
- Venture capitalists
- Value-Added Resellers (VARs)
Market Segment
This study forecasts revenue at the United Kingdom, regional, and country levels from 2020 to 2035. Decision Advisors has segmented the US Amniotic Membrane Market based on the below-mentioned segments
US Amniotic Membrane Market, By Type
- Cryopreserved Amniotic Membrane
- Dehydrated Amniotic Membrane
US Amniotic Membrane Market, By Application
- Wound Care
- Ophthalmology
- Orthopedics
- Surgical Applications
- Others
US Amniotic Membrane Market, By End User
- Hospitals
- Specialty Clinics
- Ambulatory Surgical Centers
Frequently Asked Questions (FAQ)
Q. How does variability in donor tissue quality impact product consistency in the US amniotic membrane market?
A. Variations in donor screening, tissue processing, and preservation methods can lead to differences in biological activity, which may affect clinical outcomes and product reliability across manufacturers.
Q. What challenges do manufacturers face in scaling up amniotic membrane production in the US?
A. Scaling production is limited by donor tissue availability, stringent regulatory requirements, and the need for specialized processing facilities, which can restrict supply despite growing demand.
Q. How does physician awareness and training influence adoption of amniotic membrane products?
A. Adoption rates are significantly influenced by clinician familiarity with product applications, as proper usage techniques and understanding of benefits are essential for optimal patient outcomes.
Q. What role do outpatient and home healthcare settings play in future market expansion?
A. Increasing shift toward outpatient care and home-based wound management is expected to create new opportunities, particularly for easy-to-use and longer shelf-life amniotic membrane products.
Q. How do reimbursement variations across US states affect market penetration?
A. Differences in reimbursement policies and insurance coverage across states can influence accessibility and adoption rates, especially in smaller clinics and outpatient facilities.
Check Licence
Choose the plan that fits you best: Single User, Multi-User, or Enterprise solutions tailored for your needs.
We Have You Covered
- 24/7 Analyst Support
- Clients Across the Globe
- Tailored Insights
- Technology Tracking
- Competitive Intelligence
- Custom Research
- Syndicated Market Studies
- Market Overview
- Market Segmentation
- Growth Drivers
- Market Opportunities
- Regulatory Insights
- Innovation & Sustainability
Report Details
| Scope | Country |
| Pages | 210 |
| Delivery | PDF & Excel via Email |
| Language | English |
| Release | Apr 2026 |
| Access | Download from this page |
